45 exempt human specimen meaning
USPS Packaging Instruction 6H | Postal Explorer "Exempt human or animal specimen" means a human or animal sample (including, but not limited to, secreta, excreta, blood and its components, tissue and tissue fluids, and body parts) transported for routine testing not related to the diagnosis of an infectious disease. Domestic Mail - USPS This definition does not include a human or animal patient specimen as defined in 10.17.2e. ... Exempt human or animal specimen means a human or animal sample (including, but not limited to, secreta, excreta, blood and its components, tissue and tissue fluids, and body parts) transported for routine testing not related to the diagnosis of an ...
Patient Specimen Description - University of California, Santa Cruz Definition: The U.S. Department of Transportation (DOT) and the International Air Transportation Association (IATA) define a patient specimen as a human or animal material collected directly from humans or animals and transported for research, diagnosis, investigational activities, or disease treatment or prevention.
Exempt human specimen meaning
Research Using Human Biological Specimens Details: At times, you may receive specimens that have identifiable information associated with them, but you do not need to keep the identifiers linked to the specimens.If you do not record the identifiers or link them to the specimens, the research may be eligible for review under exempt category #4. Specifically, exempt category #4 applies to research that involves the collection or study ... What does the term "exempt" actually mean in human subjects research ... Human subjects research that is classified as "exempt" means that the research qualifies as no risk or minimal risk to subjects and is exempt from most of the requirements of the Federal Policy for the Protection of Human Subjects, but is still considered research requiring an IRB review for an exemption determination. Shipping Biological Substances FAQs | UPS - United States What is an Exempt Human Specimen or Exempt Animal Specimen? "Patient specimens for which there is minimal likelihood that pathogens are present." (Dangerous Goods Regulations, 3.6.2.2.3.8) Refer to 49 USC § 173.134 for additional information regarding domestic shipping.
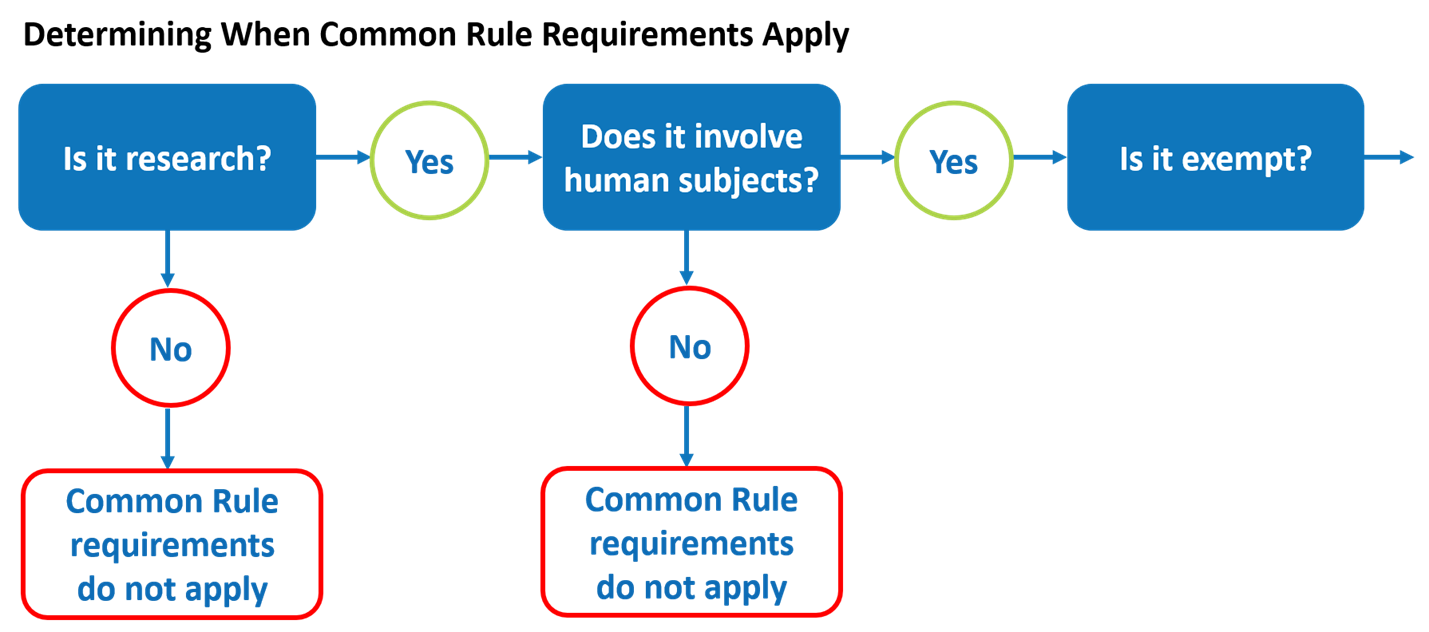
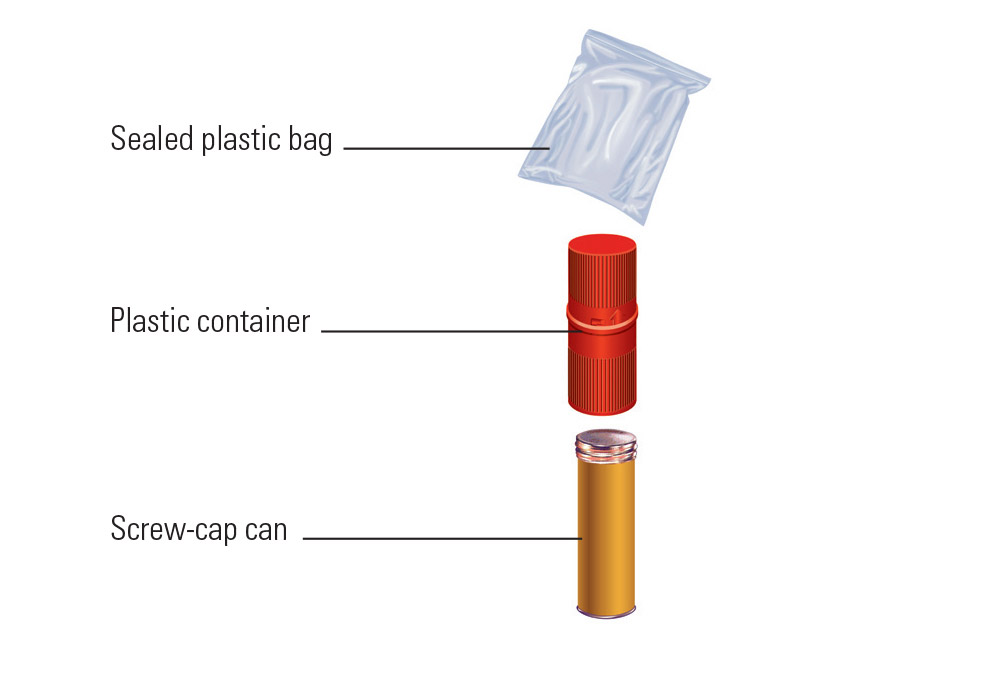
Exempt human specimen meaning. Exempt Animal or Human Specimens | Environment, Health and Safety Exempt Animal or Human Specimens Patient specimens (containing no other hazardous materials) for which there is minimal likelihood that pathogens are present are not subject to other shipping regulations except: The specimen must be packed in a packaging which will prevent any leakage and which is marked with the words: "Exempt Human Specimen", or IATA Dangerous Goods Regulations | IATA Requirements | Therapak The Exempt Human Specimen (EHS) category has a specific packing and marking requirement. Specimen shipping packages consigned to couriers and air carriers must have the marking "Exempt Human Specimen" and must, at a minimum, meet the following package requirements: 100 mm² dimension on one side leak-proof primary container PDF Exempt Human Specimen / Exempt Animal Specimen Reference Guide (IATA 3 ... Package is marked with the words "Exempt human specimen" or "Exempt animal specimen", as appropriate. (this would be in lieu of a UN3373 label). 2. The packaging must consist of three components: a. a leak-proof primary receptacle(s); b. a leak-proof secondary packaging; and PDF Packaging Requirements to Transport Biological Substances & Hazardous ... • Exempt Human Specimens • Exempt Animal Specimens The specimens collected including, but not limited to, excreta, secreta, blood and its components, tissue and tissue fluid swabs, and body parts being transported for purposes such as research, diagnosis, investigational activities, disease treatment, or prevention.
How to Ship Clinical Samples | FedEx For the purposes of this guide, clinical samples are generally defined as non-infectious human or animal materials including, but not limited to, excreta, secreta, tissue and tissue fluids, blood and FDA-approved pharmaceuticals that are blood products. PDF Step 3: Packing Category A and B and Exempt Human and Exempt Animal ... Step 3: Packing Category A and B and Exempt Human and Exempt Animal Specimens Job Aid . Use the pages below as a reference for packing Category A, B, and Exempt Specimens. Category A Substance Packaging . NOTE: The packaging is the same for both types (UN 2814 and UN2900) of Category A packaging, only the UN mark and Proper Shipping Names change. Frequently Shipped Biological Material and Proper Classification Exempt human/animal specimens are specimens in which it is NOT LIKELY that a pathogen is present. Professional judgment must be used; if you suspect the specimen may contain an infectious substance, it must be shipped accordingly. How to Ship Clinical Samples | FedEx For your convenience, we offer the FedEx Small Clinical Pak, FedEx Large Clinical Pak, FedEx ® Medium Clinical Box, and the FedEx ® Large Clinical Box as packaging options for exempt clinical-sample shipments. We recommend the FedEx Clinical Paks for use when the sturdy outer packaging of your properly packaged shipment is smaller than the minimum acceptable package size 7" x 4" x 2" (17.78 ...
PDF Regulated and Non-regulated Biological Materials Exempt Human and Animal specimens are considered dangerous goods by IATA until they meet three criteria: 1. A professional determination has been made that the material has a "minimal likelihood:" of containing any pathogens (Need Appropriate Training to meet this criteria) PDF Guidelines for Human Biospecimen - National Institutes of Health Definition of Human Biospecimens These guidelines apply to human biospecimens , including --but are not limited to --blood and other body fluids, tissues, and other biological material s obtained from humans. Subsets of human materials, such as derived cell lines that are traceable to a human subject or patients Exempt specimens - IATA Regulations - un3373.it 1. a leak-proof primary receptacle (s); 2. a leak-proof secondary packaging; and 3. an outer packaging of adequate strength for its capacity, mass and intended use, and with at least one surface having minimum dimensions of 100 mm × 100 mm; Category B - UN3373.com Exemptions: - Substances which do not contain infectious substances or substances which are unlikely to cause disease in humans or animals are not subject to these Regulations unless they meet the criteria for inclusion in another class. - Substances containing microorganisms which are non-pathogenic to humans or animals are not
Definition of Human Subjects Research | grants.nih.gov According to 45 CFR 46 , a human subject is "a living individual about whom an investigator (whether professional or student) conducting research: Obtains information or biospecimens through intervention or interaction with the individual, and uses, studies, or analyzes the information or biospecimens; or
Guidelines for the Transportation of Clinical Human Samples If the human or animal sample or biological product meets the definition of regulated medical waste in paragraph (a) (5) of this section, it must be offered for transportation and transported in conformance with the appropriate requirements for regulated medical waste.
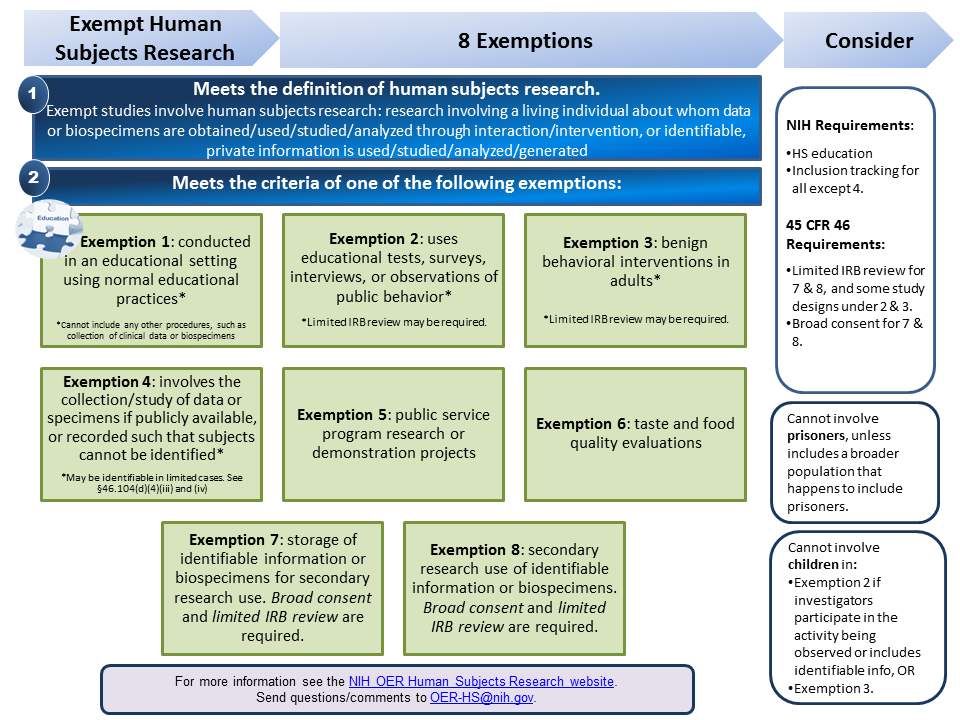
PDF 1 Meets the definition of human subjects research. specimens if publicly available, or recorded such that subjects cannot be identified* *May be identifiable in limited cases. See §46.104(d)(4)(iii) and (iv) Exemption 5: public service program research or demonstration projects Exemption 6: taste and food quality evaluations Exemption 7: storage of identifiable information or
PDF SSR-Faculty18111916510 - Marcus Institute for Aging Title: SSR-Faculty18111916510 Created Date: 11/19/2018 4:51:52 PM
IATA Training Flashcards | Quizlet The trainee's name. A description, copy, or the location of the training materials used. The month the training or exam was completed. Evidence an exam was successfully completed. What are the layers of triple packaging? (More than one answer may be correct, select all correct answers) Outer packaging.
What is "Exempt" Human Subject Research, And What Does It Mean? (2019 ... Briefly, research is termed "Exempt" when it constitutes research with human subjects, but ALSO meets the requirements of a defined low-risk category that is exempt from SOME (but not all) of the requirements governing human subjects research. To qualify as Exempt, the research must:
Exempt patient specimens - un3373.it EXEMPT HUMAN SPECIMEN or EXEMPT ANIMAL SPECIMEN They are collected directly from humans or animals and there is minimal likelihood that pathogens are present. An element of professional judgment is required to determine if a substance is exempt under this paragraph.
PDF 3 For the purposes of these Regulations - International Air Transport ... subject to these Regulations unless they meet the criteria (a) The specimen must be packed in a packaging which for inclusion in another class. will prevent any leakage and which is marked with the words "Exempt human specimen" or "Exempt 3.6.2.2.3.3 Substances in a form that any present patho- animal specimen," as appropriate;
Specimen Definition & Meaning - Merriam-Webster specimen: [noun] an individual, item, or part considered typical of a group, class, or whole. a portion or quantity of material for use in testing, examination, or study.
Exemption Categories | Research at Brown | Brown University Exemption Categories. Exempt human subjects research is a subset of minimal risk research involving human subjects that does not require approval by an IRB; however, it does require a review and a final determination by a member of the Human Research Protection Program (HRPP). The categories of Exempt research are defined by federal regulations.
Shipping Biological Substances FAQs | UPS - United States What is an Exempt Human Specimen or Exempt Animal Specimen? "Patient specimens for which there is minimal likelihood that pathogens are present." (Dangerous Goods Regulations, 3.6.2.2.3.8) Refer to 49 USC § 173.134 for additional information regarding domestic shipping.
What does the term "exempt" actually mean in human subjects research ... Human subjects research that is classified as "exempt" means that the research qualifies as no risk or minimal risk to subjects and is exempt from most of the requirements of the Federal Policy for the Protection of Human Subjects, but is still considered research requiring an IRB review for an exemption determination.
Research Using Human Biological Specimens Details: At times, you may receive specimens that have identifiable information associated with them, but you do not need to keep the identifiers linked to the specimens.If you do not record the identifiers or link them to the specimens, the research may be eligible for review under exempt category #4. Specifically, exempt category #4 applies to research that involves the collection or study ...

















Post a Comment for "45 exempt human specimen meaning"